Digitize your EHS operations with Chemical Safety Software
A Comprehensive Suite of EH&S Software Applications
TRUSTED BY




EH&S Solutions
Tightly integrated environmental health and safety software modules make it easier to achieve your objectives in less time.
Save money by purchasing only the modules that you need!
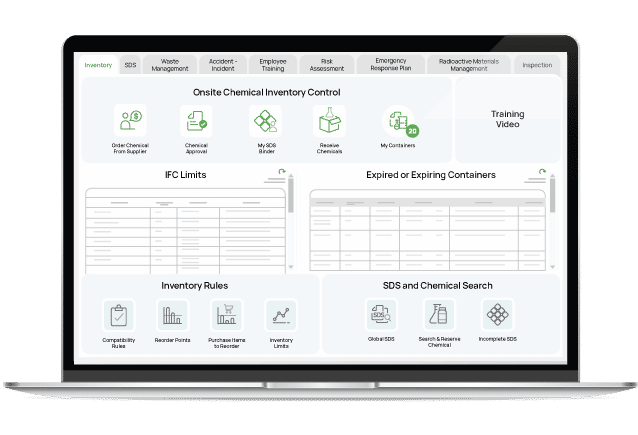
Safety Data Sheets
Maintain your collection of safety data sheets in an easily accessible cloud-based web application.
Learn more
GHS Labeling
Print GHS compliant labels and secondary container labels in a multitude of sizes and languages.
Chemical Inventory
Container tracking with barcoding and mobile apps. Track chemical inventory from delivery through to disposal and everything in between.
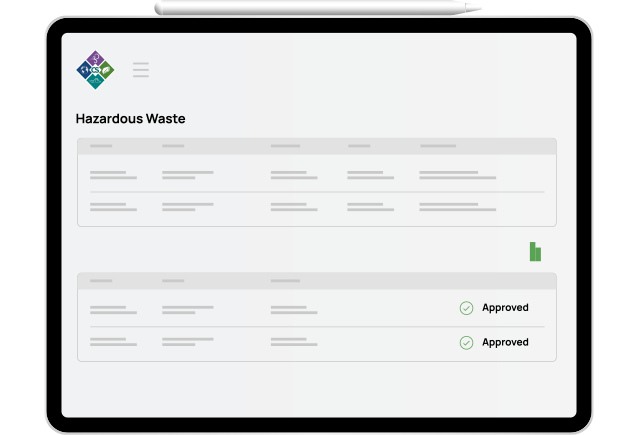
Hazardous Waste
Keep track of Haz-Waste from Cradle-to-Grave. Workflows for generation, transportation, treatment, storage, and disposal.
Environmental Regulatory Reporting
File Tier II, DHS, Biennial, IFC, VOC, HMBP reports, and many others. Stay in compliance with the latest federal, state, and local environmental regulatory requirements.
Risk Management
Perform risk assessments and Identify, prioritize, implement and monitor activities to ensure risk prevention.
Facility Safety and Compliance
Create, manage, and monitor facilities from the top down. Automate a variety of tasks that simplify information processing and reporting.
Employee Workplace Safety
Manage and track all aspects of environmental health and safety employee and workplace safety.
Radioisotope Tracking
Manage the flow of radioisotopes with real-time decay in a multi-user and multi-facility environment.
Free Add-on Mobile applications
Manage the Entire Chemical Lifecycle
Everything you need to track your chemical inventory and hazardous waste from beginning to end. EMS addresses all of the
special issues associated with storing, using, and disposing of hazardous materials.
Safety Data Sheets
Manage and distribute SDSs online.
Maintain a current library of SDS records with
easy online access for all employees.
Learn more
Chemical Inventory
Container Tracking with RFID technology, barcoding, and mobile apps.
Track chemical inventory from delivery through to disposal
and everything in between.
Learn more
Hazardous Waste
Address all of the phases of hazardous waste management in compliance with RCRA guidelines for generation, pickup, consolidation, treatment, manifesting, disposal, and reporting.
Learn more
Workplace Safety
Manage and track all aspects of environmental health and safety employee and workplace safety. Perform safety audits, inspections, training, and much more.
Learn more
Regulatory Reports
File Tier II, DHS, Biennial, IFC, VOC, HMBP reports, and many others.
Stay in compliance with the latest federal, state, and local environmental regulatory requirements.
Learn more
Why Choose Chemical Safety Software?
With more than 32 years of experience Chemical Safety Software is committed to offering innovative solutions
that prioritize the health and safety of employees and the environment.
Explore Our Blog for Industry Insights and Best Practices
Our industry-leading experts share their knowledge to help you stay ahead of the curve
TRI Reporting: What You Need to Know
TheEPA Toxics Release Inventory (TRI) Reporting program provides information on any industrial or federal facility…
SARA Tier II Reports: March 1 Deadline Rapidly Approaching
With March 1 right around the corner, it’s time to make sure your organization is…
Chemical Safety: What’s in Store for 2024?
As we are at the beginning of 2024, we at Chemical Safety Software want to…
EH&S Software Customers
Many of the world’s leading organizations rely on Chemical Safety’s EMS software to help manage their EHS operations.
Our customers are both large and small organizations, and they come from a wide variety of industries.